Certifications
MERLIN CARES FOR A CONSTANTLY HIGH QUALITY
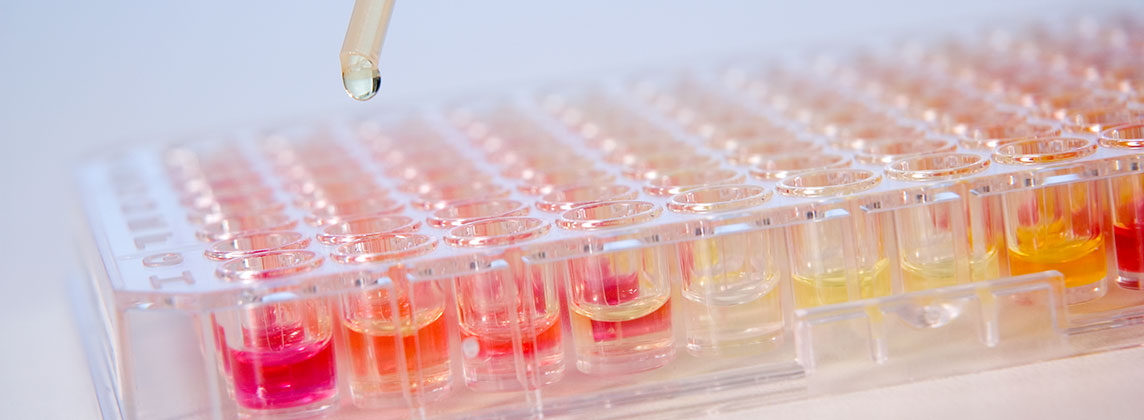
The micro plates are produced, labeled and packaged completely under clean room conditions. The process chain is completely controlled – from good receipt till final packaging. Highest quality and product performance is guaranteed due to in-process-, hygiene- and function controls for each lot.
Certified according to DIN ISO 13485
Our global quality management system is governed according to DIN ISO 13485, proven by the auditing firm TUEV SUED.
Marked with IVD-CE
All human medicine diagnostics are marked with IVD-CE according to the European guidelines.